What were the top pharma news stories as of February 2026?
Three major pharmaceutical and health technology stories shaped coverage in January 2026: a legal challenge to revised U.S. childhood vaccine guidance, a high-profile AI partnership between Bristol Myers Squibb and Microsoft to improve lung cancer detection, and OpenEvidence’s presentation of an agentic AI–driven vision for “medical super-intelligence” at the J.P. Morgan Healthcare Conference.

Februray 2026 Pharma News Breakdown
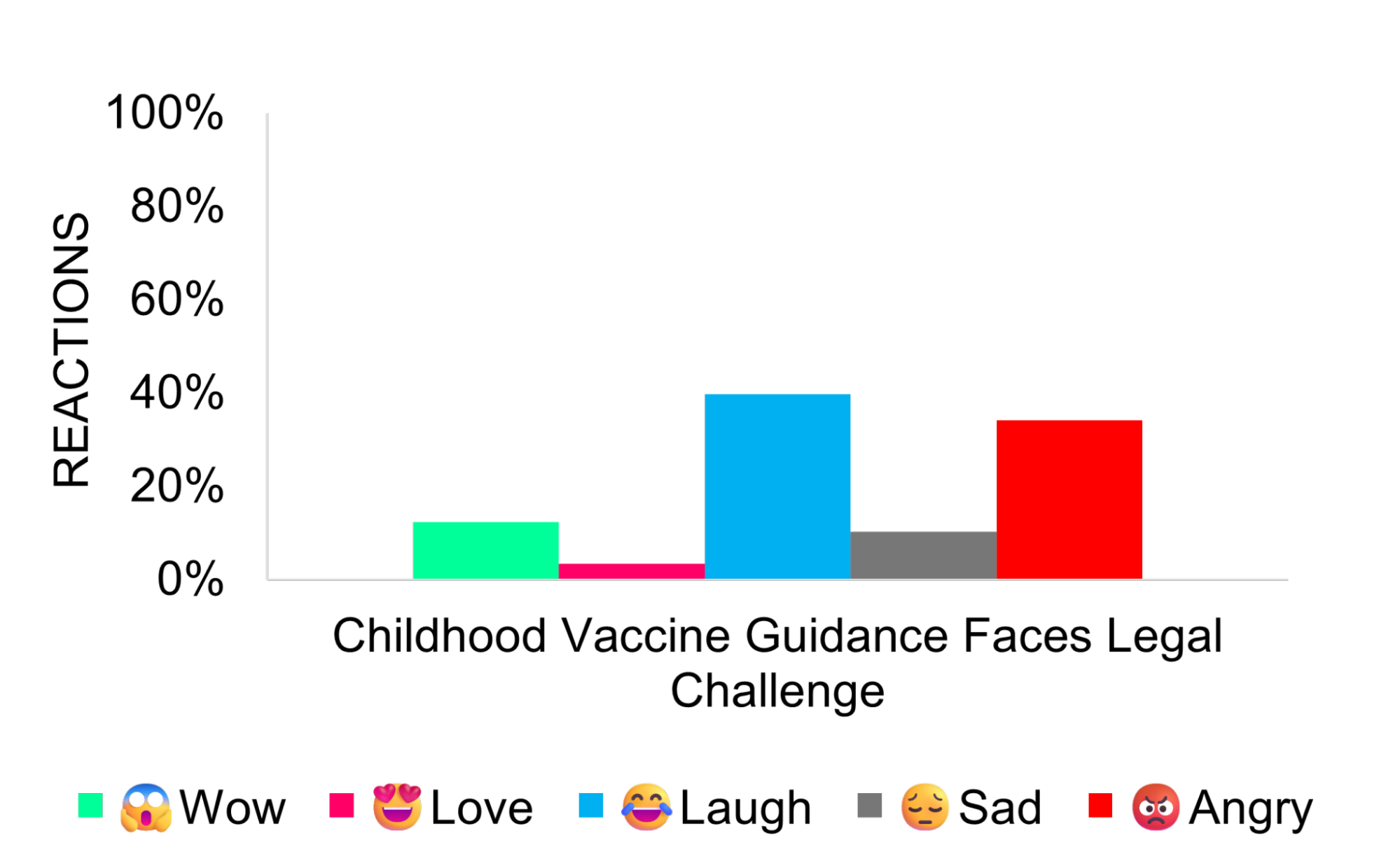
Childhood Vaccine Guidance Faces Legal Challenge
What was the lawsuit about?
Several public health organizations, led by the American Academy of Pediatrics, filed a lawsuit seeking to overturn revised CDC guidance that narrowed routine childhood vaccine recommendations. The plaintiffs alleged that Health Secretary Robert F. Kennedy Jr. improperly influenced the Advisory Committee on Immunization Practices (ACIP).
The revised guidance:
- Limited routine recommendations for certain vaccines, including influenza, rotavirus, meningococcal disease, and hepatitis A
- Shifted vaccination decisions toward physician discretion or high-risk populations
- Raised concerns regarding advisory committee balance and independence
Media coverage remained largely neutral, focusing on legal and policy implications. Supporters cited physician discretion and alignment with international practices, while critics warned the revisions could reduce clarity for families and affect public health outcomes.
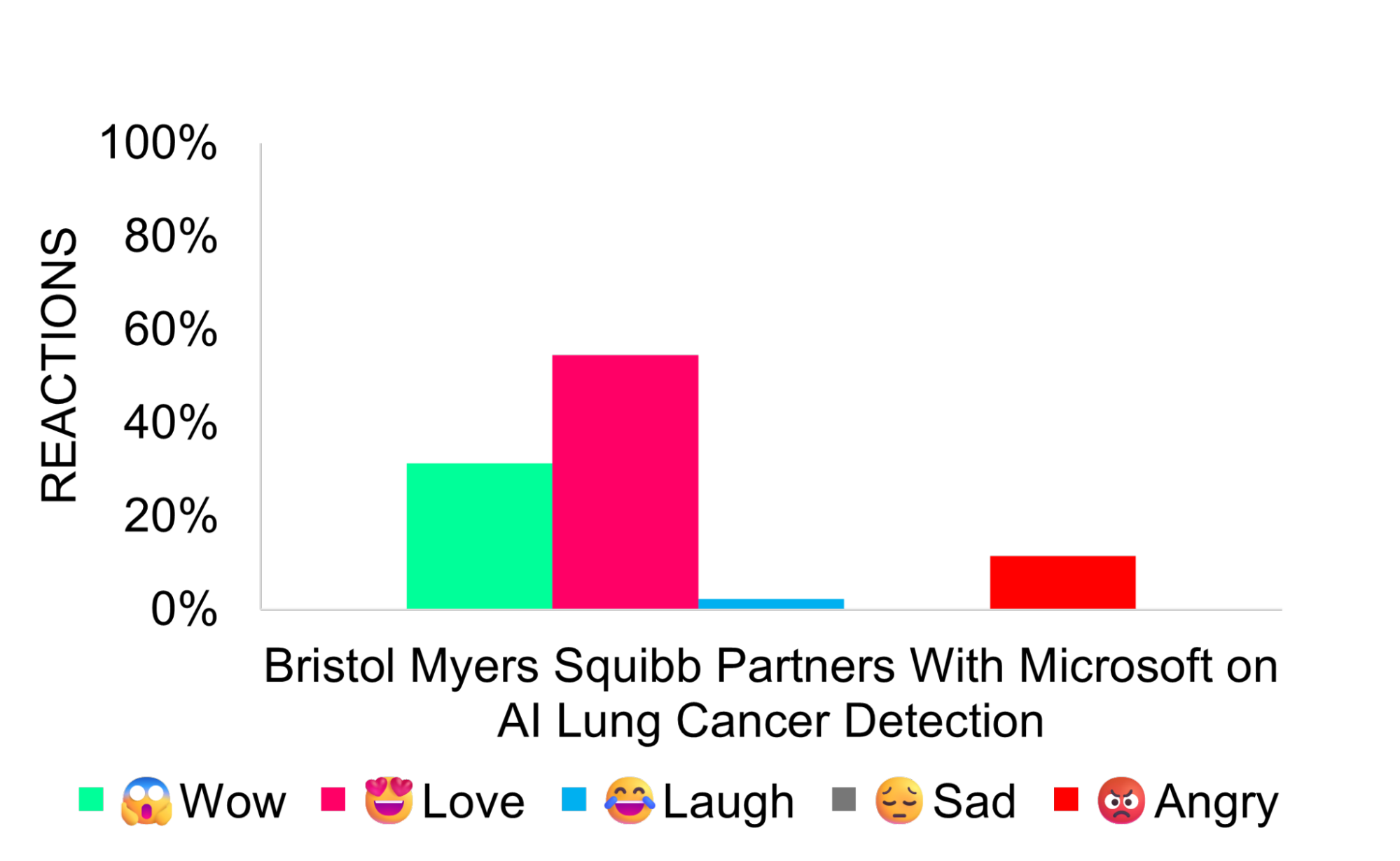
Bristol Myers Squibb Partners With Microsoft on AI Lung Cancer Detection
What is the collaboration about?
Bristol Myers Squibb signed an agreement with Microsoft to use AI-enabled radiology tools to support earlier lung cancer detection. The collaboration deploys FDA-cleared radiology AI algorithms through Microsoft’s Precision Imaging Network to analyze X-ray and CT images across U.S. hospitals.
The initiative:
- Uses artificial Intelligence tools to help clinicians identify difficult-to-detect lung nodules
- Seeks to identify patients at earlier stages of lung cancer
- Focuses on expanding access in medically underserved and rural communities
Media coverage was largely positive, emphasizing the partnership’s potential to improve early detection, enhance patient outcomes, and demonstrate how AI can advance oncology and drug development efforts.
OpenEvidence Unveils a Vision for AI-Driven Medical Super-Intelligence at JPM26
How is OpenEvidence redefining clinical intelligence with agentic AI?
At the 2026 J.P. Morgan Healthcare Conference, OpenEvidence outlined its vision for “medical super-intelligence,” describing agentic, multimodal AI systems designed to function as clinical subspecialist support tools. The company stated that its platform supports evidence-based decision-making through an AI-powered medical search engine and generative chatbot used by physicians across the United States.
Key elements include:
- Positioning agentic AI as a subspecialist support across multiple clinical domains
- Use of multimodal, multicloud architecture for scalable deployment
- Reported physician adoption across hospitals and outpatient settings
- Differentiation through partnerships with established medical journals
Media coverage was uniformly positive, highlighting OpenEvidence’s rapid growth, strong physician uptake, trusted content partnerships, and its emerging leadership role in shaping the future of AI-enabled clinical decision support.

Key Pharmaceutical Industry Takeaways for February 2026
What do these developments signal for the health and life sciences industry?
- Vaccine policy remains unsettled, with legal challenges highlighting uncertainty around federal guidance, advisory processes, and physician-led decision-making.
- AI adoption continues to expand across pharmaceutical and healthcare applications, including decision support, disease detection, and trial operations.
- Partnerships between drug manufacturers and technology companies are increasingly playing a role in improving care delivery and access.
- The industry navigates innovation alongside public trust and regulatory scrutiny amid ongoing policy and technology shifts.
Want to catch up on what shaped the industry last month?
You can also read our Top Pharma News January 2026 for context on the trends, policy shifts, and AI developments leading into February.
About Fullintel’s Pharmaceutical News Coverage
The Fullintel Hub delivers daily pharmaceutical news analysis and insights to support PR and communications teams. Our human analysts track key developments across traditional media, social platforms, and industry publications. Our award-winning methodology, including our proprietary Media Impact Score and Trust Score, helps organizations refine messaging and benchmark against competitors.
About Fullintel Strategic Media Analysis
Fullintel’s Strategic Media Analysis measures the true impact of PR and communications efforts for pharmaceutical and healthcare organizations. Angela Dwyer, Head of Insights, leads our pharmaceutical analysis practice. Her original trust factor research has been presented at PRSA ICON and IPR conferences, and she has been named a PRNEWS Top Women Award recipient and 2023 AMEC Award winner.









