What were the top pharma news stories in January 2026?
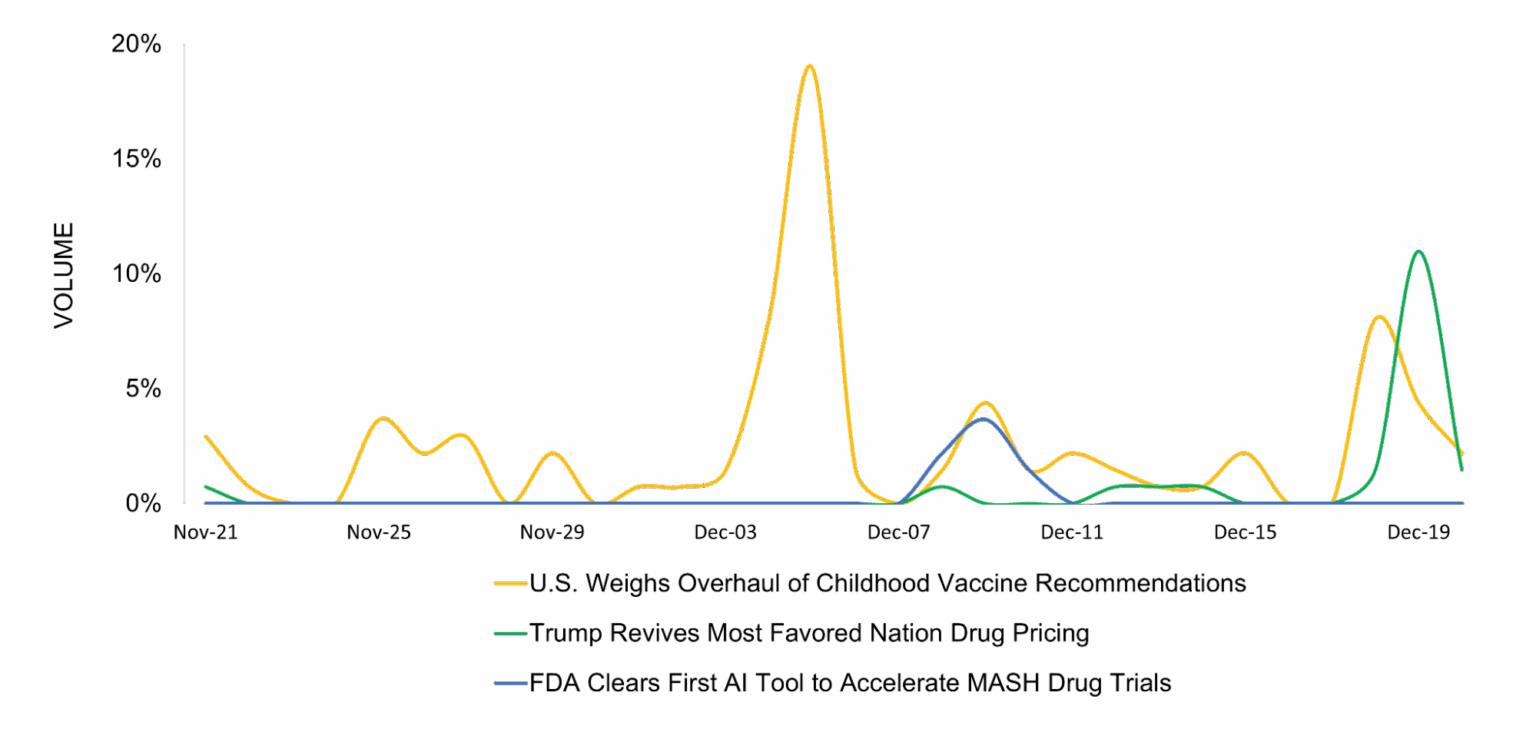
Three major pharmaceutical news stories dominated December 2025: the Trump administration’s proposed overhaul of childhood vaccine recommendations, voluntary drug pricing agreements under the Most Favored Nation policy, and the FDA’s qualification of AIM-NASH, the first AI tool for MASH clinical trials.
January 2026 Pharma News Breakdown
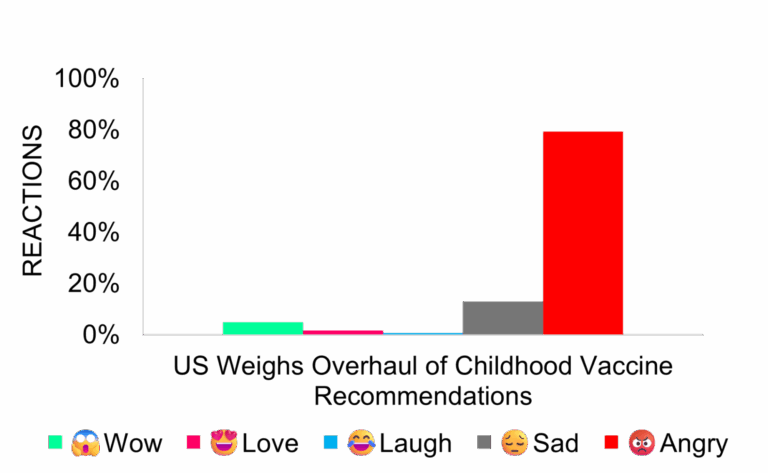
Childhood Vaccine Recommendations Face Major Overhaul
What changes are proposed for childhood vaccines?
The Trump administration is reviewing a plan to reduce federal vaccine recommendations and shift decision-making to physicians. The proposal would align the U.S. schedule more closely with European models, such as Denmark’s approach.
- End direct federal recommendations for most routine vaccines
- Reduce the total number of recommended vaccinations
- Increase physician authority in vaccination decisions
Media coverage remained neutral as details are still under review. Supporters cite parental choice and individualized care. Critics warn of reduced public health protections and potential confusion about vaccine safety.
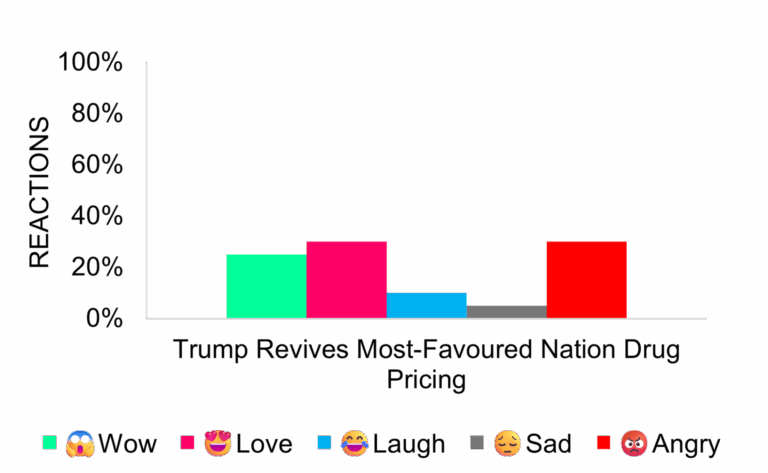
Most Favored Nation Drug Pricing Returns
Which pharmaceutical companies signed drug pricing agreements?
Several major pharmaceutical companies entered voluntary pricing agreements on December 19, 2025, under President Trump’s revived Most Favored Nation policy to lower prices for Medicaid and cash-pay drugs.
What pharmaceutical companies receive in exchange:
- Three-year exemption from pharmaceutical tariffs
- Priority FDA review scheduling
- Regulatory certainty for 36 months
Industry analysts don’t expect a material revenue impact. Many discounted drugs already carry rebates or face generic competition. Coverage increased mid-December, but sentiment stayed neutral. Questions remain about transparency and actual savings for patients.
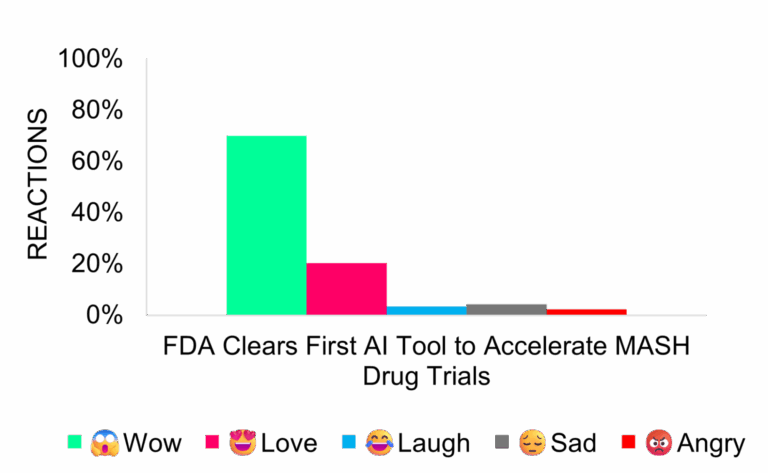
FDA Qualifies First AI Tool for MASH Clinical Trials
What is AIM-NASH, and why does it matter for pharmaceutical development?
The FDA qualified AIM-NASH on December 8, 2025, as its first AI-based tool for the development of drugs for metabolic dysfunction-associated steatohepatitis (MASH). The qualification aims to standardize liver biopsy assessments and reduce the timelines of clinical trials.
How AIM-NASH works in pharmaceutical trials:
- Analyzes biopsy images for fat, inflammation, and fibrosis
- Pathologists conduct final interpretation and validation
- Performance matches expert pathologist review in FDA testing
- Not approved for diagnostic use outside clinical trials
Today’s pharma news includes broader adoption of AI to reduce trial costs and variability. The December 8 FDA announcement generated neutral coverage focused on technical and regulatory details. Some social media responses showed enthusiasm for the FDA’s integration of AI.
Key Pharmaceutical Industry Takeaways
What do these pharmaceutical news stories mean for the industry?
- Policy uncertainty continues around vaccine recommendations and government intervention in healthcare decisions.
- Drug pricing remains a political priority, but its practical impact on pharmaceutical revenues appears to be limited.
- FDA embraces AI tools for clinical trials, potentially accelerating drug development timelines
- Pharmaceutical companies face ongoing pressure to strike a balance between affordability and innovation.
About Fullintel’s Pharmaceutical News Coverage
The Fullintel Hub delivers daily pharmaceutical news analysis and insights to help PR teams stay ahead of industry developments. Our human analysts track top pharma news across traditional media, social platforms, and industry publications.









