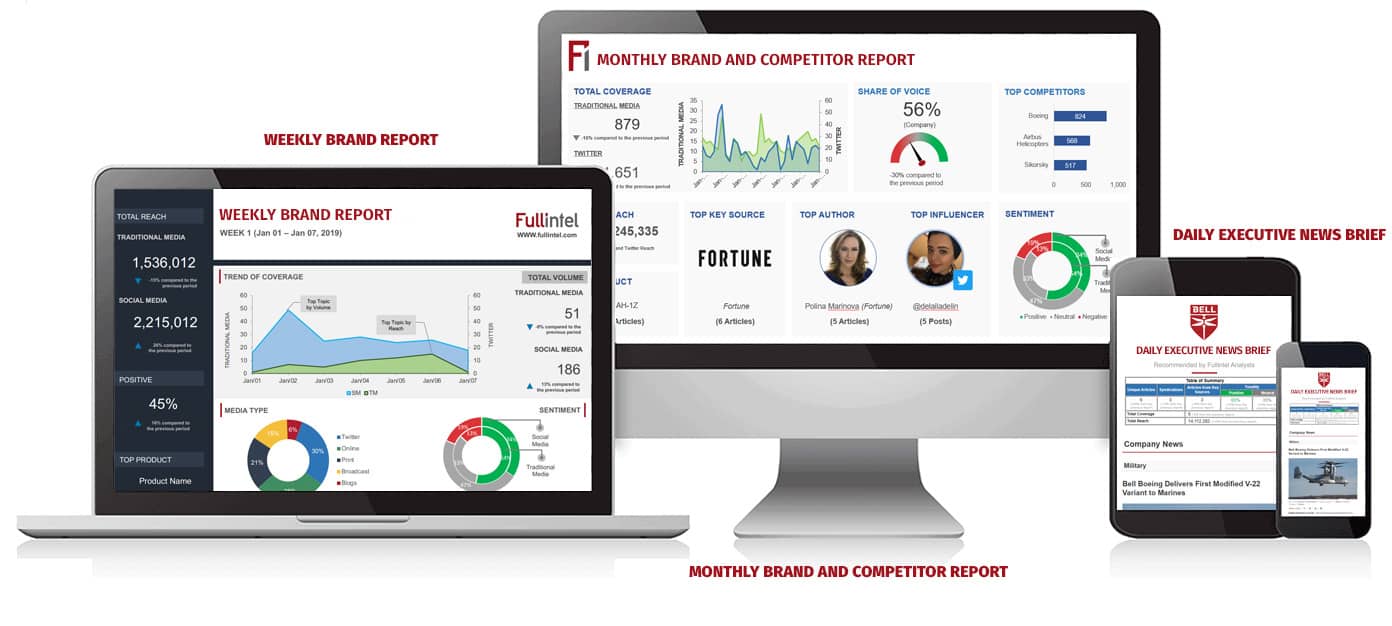
Blog
AI and Media Intelligence in 2025: How Real-Time Analytics is Reshaping PR Strategy
Artificial intelligence and sophisticated media monitoring capabilities profoundly transform the public relations landscape. As communications professionals navigate increasingly complex media environments, integrating AI-driven analytics has become advantageous and...
What We Got Right and Wrong in 2024: A PR and Communications Recap
I’m Angus, and as I look back at 2024, it’s time to review where we nailed our forecasts and where our predictions fell short. When we began the year, we laid out key trends we anticipated would reshape the PR and communications landscape. Here’s an honest assessment...
How Will Americans Deal with a 25% Increase on Avocado Toast?
Imagine starting your day with your usual avocado toast and realizing it's suddenly 25% more expensive. A culinary inconvenience for some, perhaps, but this simple scenario highlights a complex issue: how international tariffs could disrupt industries, economies, and...
Top Pharma News in November 2024
This month, the pharmaceutical industry evolves through technological innovation, regulatory oversight, and government policy. Regulatory agencies like the FDA critically shape drug development, determining market entry and research investments. Pharmaceutical...
Fullintel’s Triumph at the 2024 AMEC Awards: A Celebration of Excellence
Introduction: A Landmark Achievement In a remarkable milestone for the media intelligence industry, Fullintel has emerged as a trailblazer at the 2024 AMEC Global Communication Effectiveness Awards, securing an extraordinary five accolades across six categories. This...
PS5 Pro Launch Analysis: Slightly Improved Experience Overshadowed by High Cost, Missing Items
The PlayStation 5 Pro (PS5 Pro) debuted to considerable anticipation and hype earlier in November, with gamers and other fans eagerly awaiting the next iteration of the iconic console (which debuted back in 1994, by the way). But while plenty of writers and reviewers...
Top Pharma News in October 2024
The pharmaceutical industry experiences significant changes driven by advancements in technology, evolving drug development strategies, and strengthened partnerships. AI, precision medicine, and optimized clinical trial processes drive more efficient and personalized...
‘Fascism’ in News Headlines: Key Insights and Trends
In recent months, fascism has become a prominent topic in American media, with coverage spanning ideologies, political movements, and societal debates. Based on our analysis of 17,000 articles from September 1 to October 30, this report reveals where and how the term...
A Field Guide to AI in PR: The Skills You Need, Best Use Cases, Ethical Concerns, and More
AI is suddenly everywhere you look, including the realm of PR—so much so that “AI fatigue” has become a real thing. But if you’re feeling that slight twinge of AI fatigue, please bear with us, because AI in PR isn’t going anywhere. The key to its effective use is...
Why Social Commentary Matters: A Deep Dive into Burger King’s Post-Birth Ad Reaction Across Traditional and Social Media
In today’s complex media landscape, understanding how campaigns are received requires more than just tracking traditional media. Brands need to monitor the full spectrum of commentary, including the rich, often personal discussions that unfold on social media...
Sign up for email updates
Get PR, media monitoring, and media intelligence tips straight to your inbox and become a better communicator.
Tweets
See it live
Schedule a time with one of our product specialists to see a live, one-on-one demo.